IOP Governance-Verordnung (GIGV) - What Manufacturers Can Expect
The new Gesundheits-IT-Interoperabilitäts-Governance-Verordnung or (IOP Governance-Verordnung, GIGV for short) will come into force on October 01, 2021. A draft bill has already been published by the German Federal Ministry of Health (BMG). It does not need to be passed by parliament.
This article will explain what the regulation requires and whether you will be affected by it.
1. Background to the IOP Governance-Verordnung (GIGV)
a) Interoperability in healthcare
The healthcare system suffers from a lack of integrated processes. This leads, for example, to:
- Higher costs, e.g., due to repeated examinations
- Time delays, e.g., due to redundant data inputs
- Risks for patients, e.g., due to incomplete or contradictory data
One necessary, but not on its own sufficient, prerequisite for integrated (i.e., cross-sector as well) processes is the interoperability (IOP) of the products and systems involved in healthcare delivery. Interoperability is the ability of a system (e.g., a medical device or a piece of software) to work with other systems.
Note:
This short article on interoperability(german) presents an interoperability model. Classification into different levels makes the differences between semantic and syntactic standards easier to understand.
b) Problems the GIGV is aiming to solve
There are now numerous interoperability standards. Gematik has published a vast number of these standards in its Vesta directory. But that alone does not solve the interoperability problem:
- Problem 1: Lack of agreement on which standard to use
If there are several or simply too many standards for a use case, this hinders interoperability. A prerequisite for homogeneity is that the providers of the information systems agree on the standards to be used.
- Problem 2: Standards are not clear
Many standards deliberately leave some degree of freedom. For example, implementers can decide for themselves which data are mandatory and which are not. Syntactic standards enable different semantic standards to be included. There is a lack of “standard standardization.”
The degree of freedom can be reduced by using “profiles.” However, it is particularly difficult for non-experts to identify the level of granularity of an interoperability standard and to understand whether it is suitable for the particular use case.
- Problem 3: Standards are not complete
Every new use case, every new business model and every new type of data source (e.g., medical device, apps) increases the probability that the required interoperability standards will be missing. There is still no one organization that has a complete overview of the requirements and prioritizes the development of new standards.
The IOP Governance-Verordnung (GIGV) aims to solve this problem.
2. The approach of the IOP Governance-Verordnung (GIGV) to solving these problems
Even if it is not clearly stated: The legislator - specifically the Federal Ministry of Health (BMG) - wants to use the GIGV to curb the proliferation of interoperability standards. A new body will be established at gematik for this purpose:
“a coordinating body to promote interoperability and open standards and interfaces, and to support processes for the coordination of requirements for interfaces in information technology systems for cross-sector information exchange in healthcare”
GIGV § 2(1)
a) Tasks of the coordinating body
The task of this coordinating body is to:
- Figure out what interoperability standards are needed
- Prioritize these needs
- Continuously specify requirements for new interoperability standards
- Recommend (or make mandatory) concrete standards
- Publish these standards on an information platform
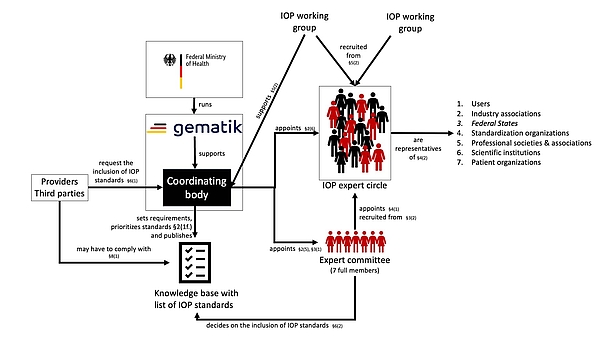
b) Interaction between the coordinating body and the expert committee, expert circle and working groups
The IOP Governance-Verordnung (GIGV) states that the coordinating body must utilize several groups of people:
- Expert committee
The coordinating body will appoint an expert committee. This committee will consist of representatives of an IOP expert circle. In addition, gematik and the BMG may each nominate one extraordinary member. Among other things, the expert committee will decide which IOP standards to include in the knowledge base. - IOP expert circle
The IOP expert circle will include experts representing various interest groups (see Fig. 1). Only people in this circle can move up to the expert committee or become a member of one of the working groups.
Ultimately, the IOP expert circle will be a group of individuals who are only capable of acting and making decisions as members of the expert committee or IOP working groups. - IOP working groups
The coordinating body will set up IOP working groups consisting of members of the expert circle to look at specific topics. The working groups will help the coordinating body to perform its tasks (see 2.a).
c) Interaction between the coordinating body and other stakeholders
External stakeholders have at least two options for interacting, indirectly at least, with the new coordinating body, expert circle and IOP working group structure:
- They can propose experts for inclusion in the “IOP expert circle” pool through associations.
- They can ask the coordinating body to add new IOP standards to the list of IOP standards on the knowledge base.
d) Manufacturers’ obligations
While §7 of the GIGV (“Recommendation of standards, profiles and guidelines for information technology systems in healthcare”) still uses the word “recommendation”, §8 is clearer.
Information technology systems in healthcare that are used in the context of the provision of health-related services or are financed in whole or in part from public funds of the Federal Ministry of Health must be designed in such a way that the recommendations made in accordance with §7 included on the information platform in accordance with §10 must be fully taken into account within 24 months of the recommendation.
GIGV § 8(1)
This means that manufacturers of medical information systems, DiGA manufacturers and certain medical device manufacturers must design the data interfaces of their products in such a way that they comply with the established IOP standards. This will affect manufacturers of hospital information systems (HIS), patient data management systems (PDMS), practice information systems (PIS) and radiology information systems (RIS).
3. Assessment of the IOP Governance-Verordnung (GIGV)
It is obviously still too early to evaluate the new GIGV. But we can make some initial observations.
a) What seems reasonable
It is obvious that there is currently a confusing array of interoperability standards. Simply listing these standards in a directory is not enough to ensure sufficient interoperability.
So, the desire for a body responsible for restoring some order is understandable.
b) Where question marks remain
However, the new regulation can also be a real headache:
- Dominance of the BMG
The GIGV follows the BMG’s current trend of imposing new regulatory requirements at short intervals while giving itself more and more power. The GIGV leaves no doubt that it will determine the IOP standards. The argument that independent experts will do this doesn’t stand up because the coordinating body will decide on all appointments to the committees and working groups. - Expertise of the experts
It is still not clear what criteria will be used to select the experts. It is clear though that the numerical distribution of various interest groups will play a role. While the optimism of standardization organizations’ representatives (for example, HL7) is justified, it remains to be seen which experts patient organizations and the Federal States are willing and able to appoint. Is interoperability state-specific? - Composition of the expert committee
This also raises the question of the make-up of the expert committee. Appointing one representative from each stakeholder group may seem democratic. However, it is manufacturers who are most affected by the definition of interoperability standards. And how is a single person supposed to represent medical technology groups, IT manufacturers and DiGA startups at the same time? How well this works (or doesn't) has been painfully demonstrated by the MDR. - Its short-term nature
As much as a quick tempo is to be welcomed, it is still hard to understand why the GIGV is being rushed through at short notice and without a significant consultation process: At the start of September 2021, there was only a draft bill. The regulation is supposed to come into force at the beginning of October.
As a result, the opportunity to request and consider feedback has been lost. Participation and active listening increase acceptance. - Overhead
It is clear that the new Governance-Verordnung doesn’t just mean additional work for gematik. Manufacturers are affected by §8 in particular. The regulation states:
“For the industry, this may result in an unquantifiable expense for the adaptation of products to uniform standards.”
Some manufacturers may find this cynical.
Everyone should be aware that a (legally enforced) change to a data interface represents a significant design change according to MDCG 2020-3. - Absence of evidence
The GIGV does not indicate what evidence it is based on. Where is the benefit-risk assessment? Where did the assumption that a two-year period for the implementation of IOP standards is the optimal balance between placing a burden on manufacturers and increasing the efficiency of the healthcare system?
The FDA is showing us how regulatory science and evidence-based regulations lead to success. Wouldn't that be an idea? The Johner Institut’s research team would help.
5. Conclusion
Unnoticed by many, the BMG has issued the IOP Governance-Verordnung (GIGV). It would be a mistake to think that this will only affect the BMG's own gematik. In fact, this regulation will also affect a lot of manufacturers of medical devices (e.g., of DiGA) and medical information systems, e.g., through new bureaucratic and additional costs for development and regulatory affairs.
Hopefully, the benefits of the new regulation will outweigh the negatives.
But it would be nicer if we didn't have to hope and already had evidence that this was the case.
Regulatory science is a branch of research that aims to formulate regulatory requirements based on evidence-based knowledge. The Johner Institut is one of many organizations conducting research on this. You can find out more on the issue of regulatory science in our
.